VIROLOGY - CHAPTER ONE
BASIC VIROLOGY: DEFINITIONS, CLASSIFICATION, MORPHOLOGY AND CHEMISTRY
Dr Margaret Hunt
Professor Emerita
Department of Pathology, Microbiology and Immunology
University of South Carolina School of Medicine
FEEDBACK
TEACHING OBJECTIVES
An introduction to viruses, their nature, structure and classification
 Figure 1
Figure 1
Relative size of viruses and bacteria Adapted from Koneman et al. Color Atlas and Textbook of
Microbiology 5th Ed. 1997 Virus images © Dr Linda Stannard,
University of Cape Town. Used with permission
 Relative
size of DNA viruses
Relative
size of DNA viruses
Images © 1995 Dr
Linda Stannard, University of Cape Town and © 1994 Veterinary Sciences Division,
Queen's University Belfast
Viruses consist of a nucleic acid (either DNA or RNA) associated with proteins
encoded by the nucleic acid. The virus may also have a
lipid bilayer membrane
(or envelope) but this is acquired from the host cell, usually by budding
through a host cell membrane. If a membrane is present, it must contain one or
more viral proteins to act as
ligands for receptors on the host cell.
Many viruses encode a few structural proteins (those that make up the mature virus particle (or virion)) and perhaps an enzyme that participates in the replication of the viral genome. Other viruses can encode many more proteins, most of which do not end up in the mature virus but participate in some way in viral replication. Herpes virus is one of the more complicated viruses and has 90 genes. Since many viruses make few or no enzymes, they are dependent on host cell enzymes to produce more virus particles. Thus, virus structure and replication are fundamentally different from those of cellular organisms. Viral dependence on the host cell for various aspects of the growth cycle has complicated the development of drugs since most drugs will inhibit cell growth as well as viral multiplication (because the same cell enzymes are used). Since a major reason to study viral metabolism is to find drugs that selectively inhibit the multiplication of viruses, we need to know when the virus uses its own proteins for part of its replication cycle - we can then try to develop drugs that inhibit the viral proteins (especially viral enzymes) specifically. In contrast to viruses, the much larger bacteria (figure 1) carry out their own metabolic processes and code for their own enzymes. Even when catalyzing similar reactions, bacterial enzymes differ from their eukaryotic homologs and can therefore be targeted by specific antibiotics. Like viruses, some bacteria (such as mycoplasma, rickettsia and chlamydia) can enter the cytoplasm of eukaryotic cells and become parasites. These small intracellular bacteria nevertheless provide all of the enzymes that are necessary for replication. Thus, mechanisms for control of bacteria, including those with a parasitic lifestyle, are more easily developed than for viruses.
|
Control measures for microorganisms include capitalizing on our knowledge of: |
||||||
|
|
Growth on artificial media |
Division by binary fission |
Whether they have both DNA and RNA |
Whether they have ribosomes |
Whether they have muramic acid |
Their sensitivity to antibiotics |
|
Bacteria |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
|
Mycoplasma |
Yes |
Yes |
Yes |
Yes |
No |
Yes |
|
Rickettsia |
No |
Yes |
Yes |
Yes |
Yes |
Yes |
|
Chlamydia |
No |
Yes |
Yes |
Yes |
No |
Yes |
|
Viruses |
No |
No |
No |
No * |
No |
No |
|
* The arenavirus family (an RNA virus family) appears to package ribosomes 'accidentally'. The packaged ribosomes appear to play no role in viral protein synthesis. |
||||||
 Relative size of positive strand RNA viruses
Relative size of positive strand RNA viruses Images © 1995 Dr Linda Stannard, University of Cape Town and © 1994 Veterinary Sciences Division, Queen's University Belfast
 Relative size of negative strand RNA viruses
Relative size of negative strand RNA viruses
Images © 1995 Dr
Linda Stannard, University of Cape Town and © 1994 Veterinary Sciences Division,
Queen's University Belfast
Figure 2. Relative sizes of DNA and RNA viruses
Viruses infect all major groups of organisms: vertebrates, invertebrates, plants, fungi, bacteria but some viruses have a broader host range than others; however, none can cross the eukaryotic/prokaryotic boundary.
Factors that affect host range include:
whether the virus can get into the host cell; that is, does it have the correct attachment protein to bind to a receptor on the cell surface? For example, HIV is largely restricted to cells that have the CD4 antigen on their surface (such as T4 cells).
if the virus can enter the cell, whether the appropriate cellular machinery is available for the virus to replicate; for example, some DNA viruses can only replicate in dividing cells which have high enough levels of deoxyribonucleotides for viral DNA synthesis.
if the virus can replicate, whether infectious virus can get out of the cell and spread the infection.
VIRUS STRUCTURE
Viruses range in size from 20 nanometers in diameter, such as the Parvoviridae, to several hundred nanometers in length in the case of the filoviridae (Figure 1 and 2).
All viruses contain a nucleic acid genome (RNA or DNA) and a protective protein coat (called the capsid). The nucleic acid genome plus the protective protein coat is called the nucleocapsid which may have icosahedral, helical or complex symmetry. Viruses may or may not have an envelope. Enveloped viruses obtain their envelope by budding through a host cell membrane. In some cases, the virus buds through the plasma membrane but in other cases the envelope may be derived from internal cell membranes such as those of the Golgi body or the nucleus. Some viruses bud through specialized parts of the plasma membrane of the host cell; for example, Ebola virus associates with lipid rafts that are rich in sphingomyelin, cholesterol and glypiated proteins. Poxviruses are exceptional in that they wrap themselves in host cell membranes using a mechanism that is different from the usual budding process used by other viruses.
Enveloped viruses do not necessarily have to kill their host cell in order to be released, since they can bud out of the cell - a process that is not necessarily lethal to the cell - hence some budding viruses can set up persistent infections.
Enveloped viruses are readily infectious only if the envelope is intact (since the viral attachment proteins which recognize the host cell receptors are in the viral envelope). This means that agents that damage the envelope, such as alcohols and detergents, reduce infectivity.
WEB RESOURCES
Principles
of virus architecture
Linda Stannard
Platonic Solids
University of Utah
Triangulation Numbers
J-Y Sgro
VIRION NUCLEOCAPSID STRUCTURES
Icosahedral symmetry
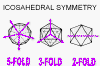
An icosahedron is a Platonic solid with twenty faces (figure 3A) and 5:3:2 rotational symmetry (figure 3B). There are six five-fold axes of symmetry through which the icosahedron can be rotated passing through the vertices, ten 3-fold axes of symmetry passing though each face and fifteen two-fold axes of symmetry passing through the edges (figure 3B). There are twelve corners or vertices and 5-fold symmetry around vertices (figure 3C). The capsid shell is made of repeating subunits of viral protein (There may be one kind of subunit or several, according to the virus). All faces of the icosahedron are identical.
VIDEO
Mathematical virology: a novel approach to the structure and assembly of
viruses
The nucleic acid is packaged inside the capsid shell and protected from the environment by the capsid (figure 3D).
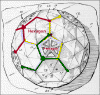
Proteins associate into structural units (this is what we see in the electron microscope or when we start to disassociate a capsid), the structural units are known as capsomers. Capsomers may contain one or several kinds of polypeptide chain. Capsomers at the 12 corners have a 5-fold symmetry and interact with 5 neighboring capsomers, and are thus known as pentons or pentamers (figure 3E). Larger viruses contain more capsomers; extra capsomers are arranged in a regular array on the faces of the icosahedrons. They have six neighbors and are called hexons or hexamers (figure 3F).
The size of an icosahedron depends on the size and number of capsomers; there will always be 12 pentons (at each corner) but the number of hexons increases with size (figure 3H). A good example of an icosahedral virus is human adenovirus which contains the usual twelve pentons plus two hundred and forty hexons (figure 3G and I). The symmetrical formation of hexagonal arrays on a flat face occurs in many situations; for example, in the packing of test tubes in a box (figure 3J). It can also be seen in the packing of the subunits of herpes virus, an enveloped icosahedral virus. In figure 3K, the external membrane of herpes simplex has been removed to reveal the nucleocapsid. Although icosahedrons are flat-faced (as in figure 3A), viral icosahedrons are usually round as seen in figure 3K. A good example of a small round icosahedron is a normal soccer ball (figure 3L). A larger icosahedron is a geodesic dome (figure 3M).
Figure 3
Viral capsid icosahedral symmetry
A Icosahedron: 20 triangular faces
C Five fold symmetry at vertices
D Nucleic acid is packaged inside the capsid
© J-Y Sgro, Used with permission. From Virus World
E Capsomers at the 12 corners have a 5-fold symmetry and interact with 5 neighboring capsomers, and are thus known as pentons (or pentamers).
F-i
Larger viruses contain more capsomers, extra capsomers are arranged in a regular array on the faces of the icosahedrons, these often have six neighbors and are called hexons
F-ii
Herpes nucleocapsid showing pentons at the vertices of the icosahedron Zhou et al. Baylor College of Medicine Reference: Z. H. Zhou, B.V.V Prasad, J. Jakana, F.R. Rixon, W. Chiu Baylor College of Medicine, Journal of Molecular BiologyG Adenovirus symmetry
H Components of an icosahedral capsid
I Human adenovirus seen by negative staining © 1995 Dr Linda Stannard, University of Cape Town. Used with permission
J Packing of uniform circular objects in a hexagonal array
K 3-D computer reconstruction from cryo-electron micrographs of herpes simplex virus capsids. Rotating image. National Institutes of Health
L The icosahedral shape of a soccer ball. Note that the ball consists of penton subunits (black) and hexon subunits (white)
Figure 4
Helical symmetry
Helical symmetry
Protein subunits can interact with
each other and with the nucleic acid to form a coiled, ribbon like structure.
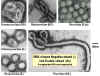
The best studied virus with helical symmetry is the non-enveloped plant virus,
tobacco mosaic virus (Figure 4 A-E). The helical nature of this virus is quite
clear in negative staining electron micrographs since the virus forms a rigid
rod-like structure. In enveloped, helically symmetrical viruses (e.g.
influenza virus,
rabies virus),
the capsid is more flexible (and longer) and appears in negative stains rather
like a telephone cord (figure 4H, I).
A Tobacco mosaic virus structure showing a helical capsid structure
B Close up of a single TMV rod. Image from the International Committee on Taxonomy of Viruses database.
C Close up of tobacco mosaic virus rods © 1994 Rothamsted Experimental Station
D Tobacco Mosaic Virus (TEM x207,480) © Dennis Kunkel Microscopy, Inc. Used with permission
E Tobacco Mosaic Virus (TEM x376,200) © Dennis Kunkel Microscopy, Inc. Used with permission
F Rabies virus Wadsworth Center, NY Dept of Health
G Influenza Virus © 1995 Dr Linda Stannard, University of Cape Town. Used with permission
H Transmission electron micrograph: A/New Jersey/76 (Hsw1N1) swine flu virus. CDC/ Dr. E. Palmer; R.E. Bates
I Graphical representation of a generic influenza virion’s ultrastructure. A portion of the virion’s outer protein coat has been cut away, which reveals the virus’ nucleocapsid. CDC/ Doug Jordan
Complex symmetry
These are regular structures, but the nature of
the symmetry is not fully
understood. Examples include the poxviruses (Figure 5).
Complex symmetry
A Complex symmetry found in poxviruses Fenner and White Medical Virology 4th Ed. 1994
B Pox virus seen by negative staining © Stewart McNulty, 1994 Veterinary Sciences Division, Queen's University Belfast Used with permission
C Molluscum contagiosum virus- a Molluscipoxvirus © 1995 Dr Linda Stannard, University of Cape Town. Used with permission
 Five basic types of virus symmetry
Five basic types of virus symmetry
Figure 6
FIVE BASIC STRUCTURAL FORMS OF VIRUSES IN NATURE
-
Naked icosahedral e.g. poliovirus, adenovirus, hepatitis A virus
-
Naked helical
e.g. tobacco mosaic virus. So far no human viruses with this
structure are known
-
Enveloped icosahedral e.g. herpes virus, yellow fever virus, rubella virus
-
Enveloped helical e.g. rabies virus, influenza virus, parainfluenza virus, mumps virus, measles virus
-
Complex e.g. poxvirus
(Figure 6)
UNCONVENTIONAL AGENTS
There are also the 'unconventional agents' sometimes known as 'unconventional viruses' or 'atypical viruses' - Up to now, the main kinds that have been studied are viroids and prions.
SATELLITES
Satellites are DNA or RNA molecules between 300 and 2000
nucleotides in length that require a helper virus for their replication. They
therefore parasitize viruses. Some satellites code for a capsid protein, and in
this case cells infected by a satellite and its helper virus release two
distinct types of virus particle – one composed of the satellite nucleic acid
associated with its capsid, the other corresponding to the helper virus. Other
satellites do not code for their own capsid protein, but use the capsid protein
of the helper virus. In this case, the virions that carry the satellite RNA or
DNA are identical, or very similar, to those of the helper virus.
VIROIDS
Viroids contain RNA only. They are small (less than 400 nucleotides), single stranded, circular RNAs. The RNAs are not packaged, do not appear to code for any proteins, and so far have only been shown to be associated with plant disease. However, there are some suggestions that somewhat similar agents may possibly be involved in some human diseases.
Hepatitis delta virus
At present, the only known human disease agent to resemble viroids is hepatitis delta virus (HDV). In some ways HDV (also called hepatitis delta agent) appears to be intermediate between 'classical viruses' and viroids. HDV has a very small RNA genome (~1700 nucleotides) compared to most viruses, although it is somewhat larger than viroids. However, features of HDV's nucleic acid sequence and structure are similar to some viroids. HDV differs from viroids in that it codes for a protein (various forms of the hepatitis delta antigen). Unlike the viroids, it is packaged. However, it differs from true viruses in that it does not code for its own attachment protein. The RNA is encapsidated by the hepatitis delta antigen, and HDV acts as a parasite on the unrelated hepatitis B virus (HBV), using HBV envelopes containing the hepatitis B attachment protein (HBsAg).
PRIONS
Prions contain protein only (although this is somewhat controversial). They are small, proteinaceous particles and there is controversy as to whether they contain any nucleic acid, but if there is any, there is very little, and almost certainly not enough to code for protein: Examples of prion-caused human diseases are Kuru, Creutzfeldt-Jakob disease and Gerstmann-Straussler syndrome. Prions also cause scrapie in sheep.
ARE VIRUSES LIVING OR DEAD?
This depends on the definition of life. To avoid possible arguments, we often refer to whether they have or have lost some aspect of their biological activities rather than referring to living or dead viruses. Hence we talk about number of infectious particles, or number of plaque forming particles rather than number of living particles.
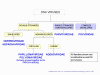 Families of DNA viruses. All families shown are icosahedral except
poxviruses
Families of DNA viruses. All families shown are icosahedral except
poxviruses
Figure 7
Modified from Volk et al., Essentials of Medical
Microbiology. 4th Ed
CLASSIFICATION OF VIRUSES
The internationally agreed system of virus classification is based on the structure and composition of the virus particle (virion) (Figure 7). In some cases, the mode of replication is also important in classification. Viruses are classified into various families on this basis.
INTERNATIONAL CLASSIFICATION OF VIRUSES
Primary characteristics used in classification
Viruses are classified according to the nature of their genome and their structure
|
VIRAL CLASSIFICATION |
|
|
Nucleic acid |
RNA or DNA |
| single-stranded or double-stranded | |
| non-segmented or segmented | |
| linear or circular | |
| if genome is single stranded RNA, can it function as mRNA? | |
| whether genome is diploid (such as in retroviruses) | |
|
Virion structure |
symmetry (icosahedral, helical, complex) |
| enveloped or not enveloped | |
| number of capsomers | |
Secondary characteristics
Replication strategy
Sometimes a group of viruses that seems to be a single group by the above criteria is found to contain a subgroup of viruses which have a fundamentally different replication strategy - in this case the group will be divided based on the mode of replication.
SOME VIRUSES OF POTENTIAL INTEREST
I = ICOSAHEDRAL SYMMETRY, H = HELICAL SYMMETRY, C = COMPLEX SYMMETRY
|
DNA VIRUSES |
|||||
|
|
Symmetry |
Envelope |
Size |
Virion polymerase |
Comments and some examples |
|
PARVOVIRIDAE |
I |
- |
20nm |
- |
Include adeno-associated virus, human parvovirus B19. |
|
HEPADNAVIRIDAE |
I |
+ |
42nm |
+ |
DNA replicates via an RNA intermediate. Includes hepatitis B virus which may increase risk of hepatocarcinoma. |
|
PAPILLOMA- VIRIDAE * |
I |
- |
40-60nm |
- |
some members cause warts, some associated with increased risk of cervical cancer |
|
POLYOMA-VIRIDAE * |
I |
- |
40-60nm |
- |
SV40, some members cause PML.
|
|
ADENOVIRIDAE |
I |
- |
80nm |
- |
More than 40 human serotypes |
|
HERPESVIRIDAE |
I |
+ |
190nm |
- |
Latency common. Includes herpes simplex type 1 and 2, varicella zoster virus (chicken pox, shingles), Epstein Barr virus (infectious mononucleosis), cytomegalovirus. |
|
POXVIRIDAE |
C |
+ |
200nm x 350nm |
+ |
Vaccinia, smallpox, cowpox viruses Cytoplasmic, very complex. |
|
* Formerly grouped together as the PAPOVAVIRIDAE |
|||||
THE ABOVE DNA VIRUS FAMILIES ARE LISTED IN ORDER OF INCREASING GENOME SIZE
|
RNA VIRUSES - POSITIVE SENSE |
|||||
|
|
Symmetry |
Envelope |
Size |
Virion polymerase |
Comments and some examples |
|
PICORNAVIRIDAE |
I |
- |
30nm |
- |
Includes enteroviruses, rhinoviruses, coxsackie virus, poliovirus, hepatitis A virus |
|
CALICIVIRIDAE |
I |
- |
35nm |
- |
gastroenteritis, Norwalk agent probably a member |
|
TOGAVIRIDAE |
I |
+ |
60-70nm |
- |
Alphavirus genus: includes western equine encephalitis virus (WEE), eastern equine encephalitis virus (EEE),Venezuelan equine encephalitis virus, Chikungunya virus, Sindbis virus, Semliki Forest virus Rubrivirus genus: contains only rubella virus |
|
FLAVIVIRIDAE |
I |
+ |
40-55nm |
- |
Include yellow fever, dengue, Japanese encephalitis, St. Louis encephalitis viruses, etc. Have only recently been given family status (formerly classed with Togaviridae). |
|
CORONAVIRIDAE |
H |
+ |
75-160nm |
- |
Estimated responsible for 10-30% of common colds |
|
RETROVIRIDAE |
I |
+ |
100nm |
+ |
Have reverse transcriptase, some members oncogenic in animals. HIV is a member. Diploid genome. |
|
RNA VIRUSES - NEGATIVE SENSE |
|||||
|
|
Symmetry |
Envelope |
Size |
Virion polymerase |
Comments and some examples |
|
RHABDOVIRIDAE
|
H |
+ |
60 x 180nm |
+ |
These include rabies virus, vesicular stomatitis virus, Mokola virus, Duvenhage virus |
|
PARAMYXOVIRIDAE |
H |
+ |
150-300nm |
+ |
Includes Newcastle disease virus, parainfluenza viruses, mumps virus, measles virus, respiratory syncytial virus |
|
ORTHOMYXOVIRIDAE |
H |
+ |
80-120nm |
+ |
Influenza type A and B viruses have segmented genome. They steal mRNA caps |
|
BUNYAVIRIDAE |
H |
+ |
95nm |
+ |
Over 86 members, most have arthropod vectors. Members include California encephalitis, LaCrosse, Crimean-Congo hemorrhagic fever, and Rift Valley fever viruses. Members of the hantavirus genus (includes agents of Korean hemorrhagic fever, human pulmonary syndrome in USA) seem to have rodent vectors. Segmented genome. |
|
ARENAVIRIDAE |
H |
+ |
50-300nm |
+ |
Includes lymphocytic choriomeningitis, Lassa, Junin (Argentine hemorrhagic fever), and Machupo (Bolivian hemorrhagic fever) viruses. Segmented genome |
|
FILOVIRIDAE |
H |
+ |
80nm x 800-900nm |
+ |
Marburg virus, Ebola virus, Reston virus |
|
RNA VIRUSES - DOUBLE STRANDED |
|||||
|
|
Symmetry |
Envelope |
Size |
Virion polymerase |
Comments and some examples |
|
REOVIRIDAE |
I |
- |
75nm |
+ |
The reoviridae include the reovirus, rotavirus and orbivirus genera. Human reovirus infections are apparently asymptomatic., Members of this group that affect humans include Colorado tick fever virus (orbivirus) and human rotaviruses (can cause gastroenteritis). All of these viruses have segmented genomes. |
Return to the Virology section of Microbiology and Immunology On-line
Return to the Home Page of Microbiology and Immunology On-line
This page last changed on Monday, July 21, 2003
Page maintained by Richard Hunt